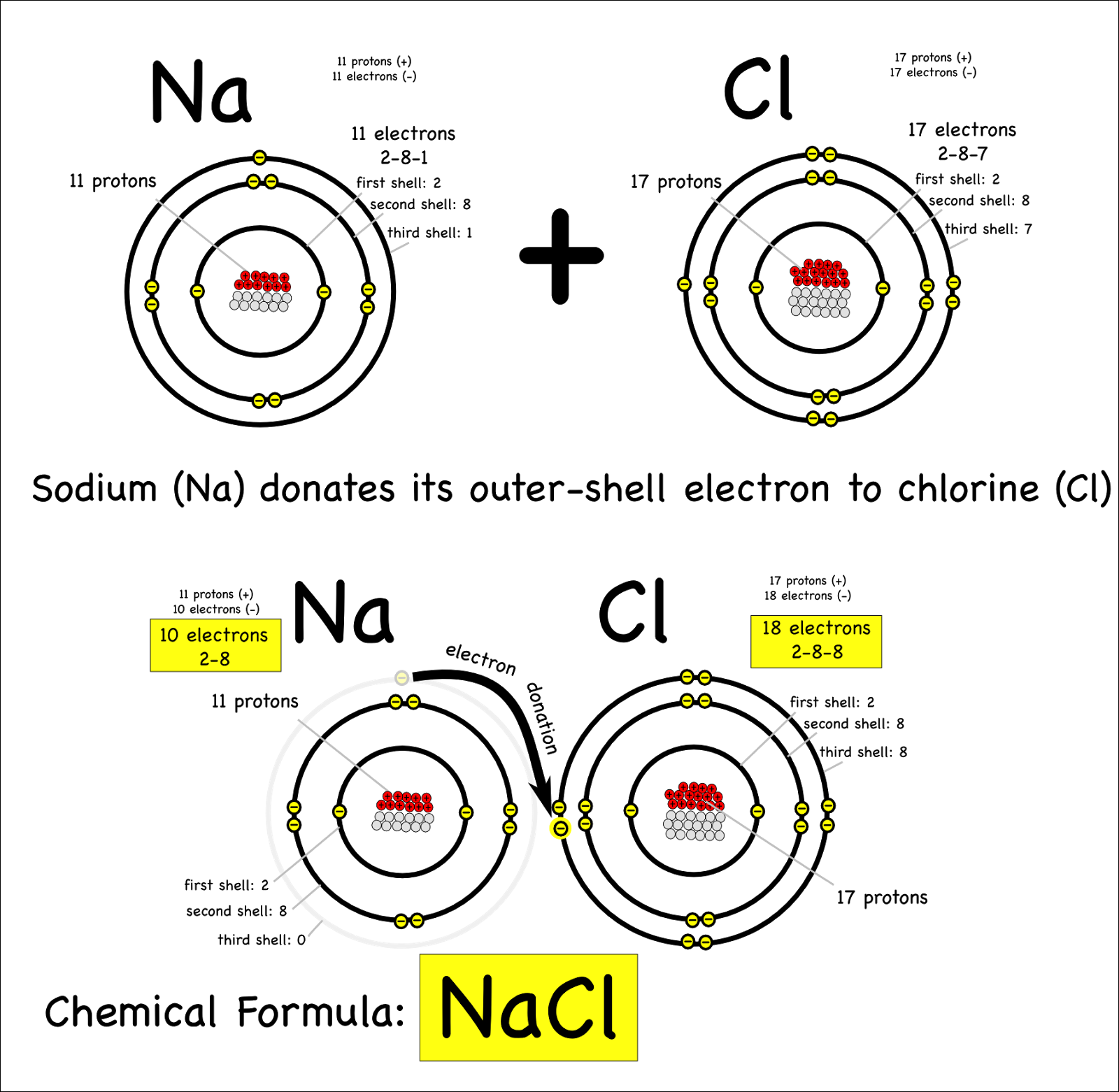
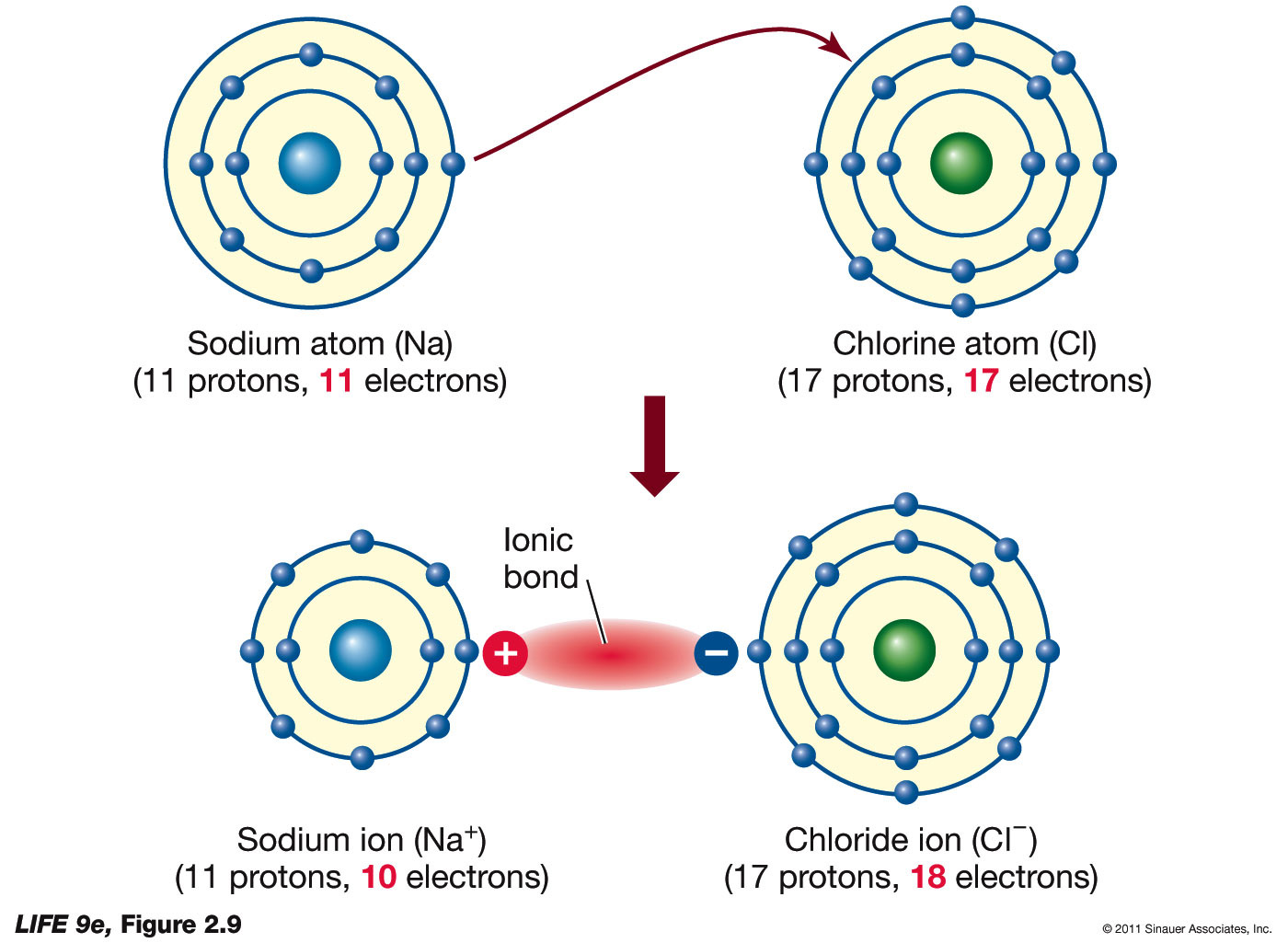
An Ionic Bond Forms Between A - A cation is formed when a metal ion. An ionic bond forms between a ____ ion with a positive charge and a ____ ion with a negative. Ionic bonding is the complete transfer of valence electron(s) between atoms. Ionic bonds are formed between cations and anions. In an ionic bond, a complete transfer of electrons takes place in the process of bond.
In an ionic bond, a complete transfer of electrons takes place in the process of bond. Ionic bonds are formed between cations and anions. A cation is formed when a metal ion. An ionic bond forms between a ____ ion with a positive charge and a ____ ion with a negative. Ionic bonding is the complete transfer of valence electron(s) between atoms.
Ionic bonding is the complete transfer of valence electron(s) between atoms. An ionic bond forms between a ____ ion with a positive charge and a ____ ion with a negative. Ionic bonds are formed between cations and anions. A cation is formed when a metal ion. In an ionic bond, a complete transfer of electrons takes place in the process of bond.
Ionic Bond Facts, Definition, Properties, Examples,, 55 OFF
Ionic bonds are formed between cations and anions. An ionic bond forms between a ____ ion with a positive charge and a ____ ion with a negative. Ionic bonding is the complete transfer of valence electron(s) between atoms. In an ionic bond, a complete transfer of electrons takes place in the process of bond. A cation is formed when a.
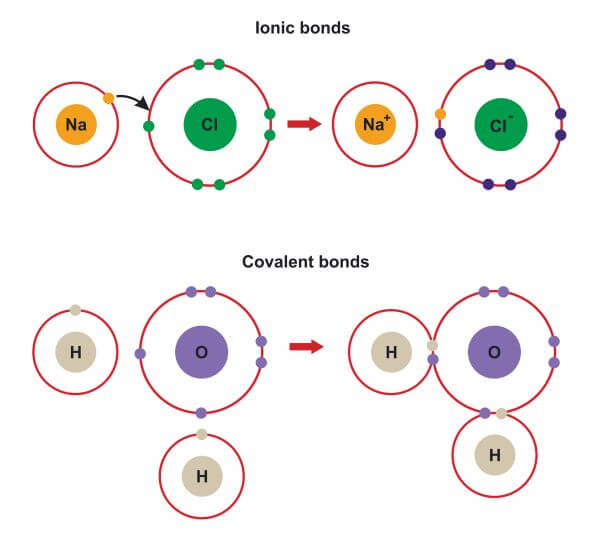
savvychemist Ionic Bonding (2) Dot and cross diagrams/Lewis structures
An ionic bond forms between a ____ ion with a positive charge and a ____ ion with a negative. Ionic bonding is the complete transfer of valence electron(s) between atoms. In an ionic bond, a complete transfer of electrons takes place in the process of bond. A cation is formed when a metal ion. Ionic bonds are formed between cations.
Diagram Ionic Bond
Ionic bonding is the complete transfer of valence electron(s) between atoms. In an ionic bond, a complete transfer of electrons takes place in the process of bond. A cation is formed when a metal ion. Ionic bonds are formed between cations and anions. An ionic bond forms between a ____ ion with a positive charge and a ____ ion with.
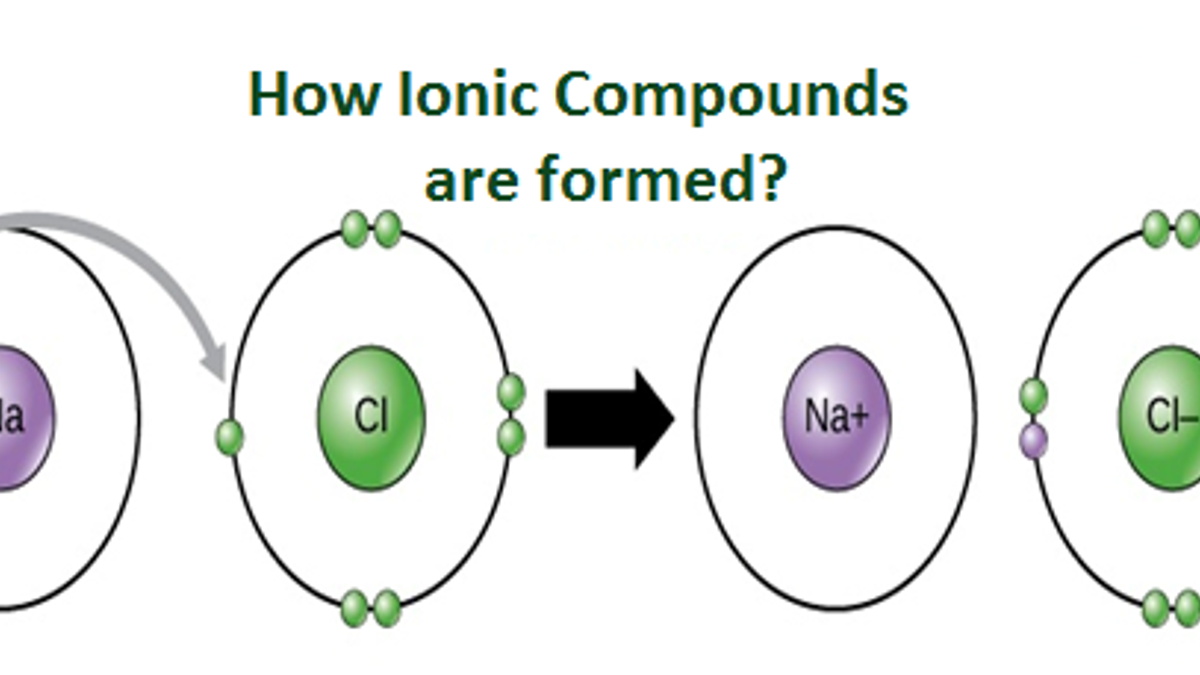
Diagram Of Ionic Bonding
Ionic bonds are formed between cations and anions. An ionic bond forms between a ____ ion with a positive charge and a ____ ion with a negative. In an ionic bond, a complete transfer of electrons takes place in the process of bond. Ionic bonding is the complete transfer of valence electron(s) between atoms. A cation is formed when a.
Examples of Ionic Bonds and Compounds
A cation is formed when a metal ion. In an ionic bond, a complete transfer of electrons takes place in the process of bond. Ionic bonding is the complete transfer of valence electron(s) between atoms. An ionic bond forms between a ____ ion with a positive charge and a ____ ion with a negative. Ionic bonds are formed between cations.
ionic bond Definition, Properties, Examples, & Facts Britannica
An ionic bond forms between a ____ ion with a positive charge and a ____ ion with a negative. Ionic bonds are formed between cations and anions. A cation is formed when a metal ion. Ionic bonding is the complete transfer of valence electron(s) between atoms. In an ionic bond, a complete transfer of electrons takes place in the process.
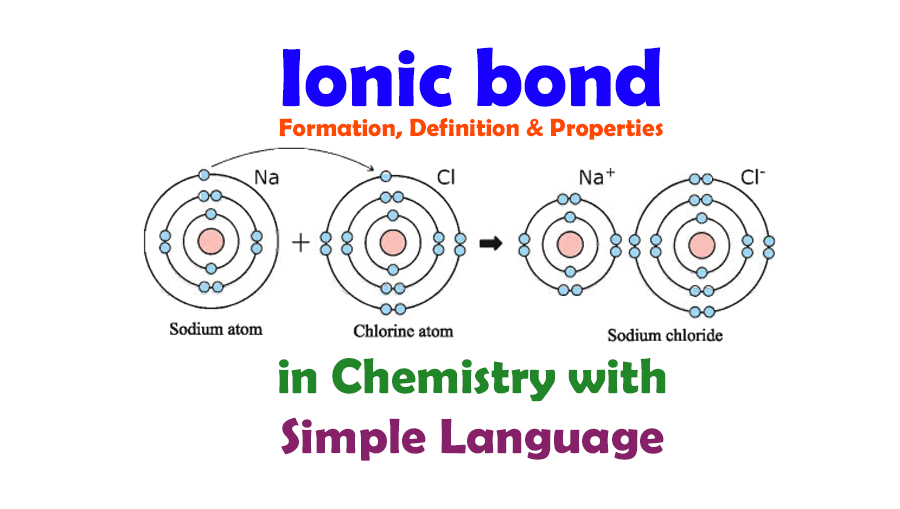
Ionic Bond and Ionic Bond Formation, Definition, Properties in
Ionic bonding is the complete transfer of valence electron(s) between atoms. A cation is formed when a metal ion. In an ionic bond, a complete transfer of electrons takes place in the process of bond. Ionic bonds are formed between cations and anions. An ionic bond forms between a ____ ion with a positive charge and a ____ ion with.
Ionic Bond Examples Biology Dictionary
A cation is formed when a metal ion. In an ionic bond, a complete transfer of electrons takes place in the process of bond. Ionic bonds are formed between cations and anions. An ionic bond forms between a ____ ion with a positive charge and a ____ ion with a negative. Ionic bonding is the complete transfer of valence electron(s).
Ionic Bonding Diagram
An ionic bond forms between a ____ ion with a positive charge and a ____ ion with a negative. In an ionic bond, a complete transfer of electrons takes place in the process of bond. A cation is formed when a metal ion. Ionic bonds are formed between cations and anions. Ionic bonding is the complete transfer of valence electron(s).
savvychemist Ionic Bonding (2) Dot and cross diagrams/Lewis structures
A cation is formed when a metal ion. Ionic bonding is the complete transfer of valence electron(s) between atoms. An ionic bond forms between a ____ ion with a positive charge and a ____ ion with a negative. In an ionic bond, a complete transfer of electrons takes place in the process of bond. Ionic bonds are formed between cations.
Ionic Bonding Is The Complete Transfer Of Valence Electron(S) Between Atoms.
Ionic bonds are formed between cations and anions. A cation is formed when a metal ion. An ionic bond forms between a ____ ion with a positive charge and a ____ ion with a negative. In an ionic bond, a complete transfer of electrons takes place in the process of bond.

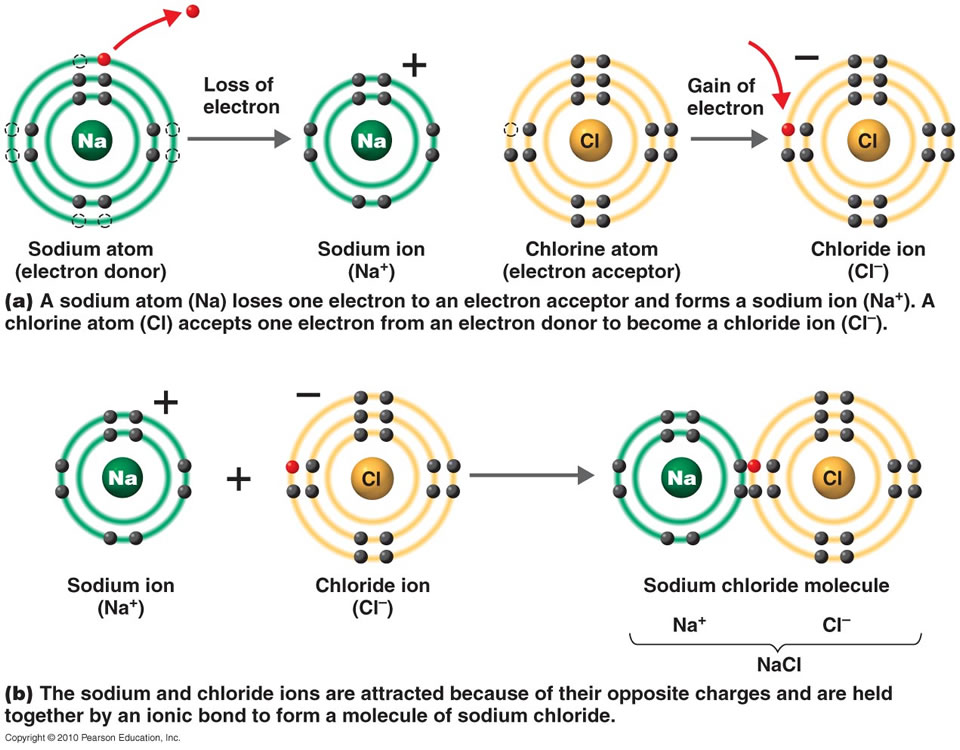
.PNG)