How Do Hydrogen Bonds Form Between Water Molecules - Hydrogen bonds form between water molecules when the partially positive end of. The covalent bonds of water make it a polar molecule. Identify three special properties of water that make it unusual for a molecule of its.
The covalent bonds of water make it a polar molecule. Identify three special properties of water that make it unusual for a molecule of its. Hydrogen bonds form between water molecules when the partially positive end of.
Identify three special properties of water that make it unusual for a molecule of its. The covalent bonds of water make it a polar molecule. Hydrogen bonds form between water molecules when the partially positive end of.
Water Molecules Hydrogen Bonds
The covalent bonds of water make it a polar molecule. Hydrogen bonds form between water molecules when the partially positive end of. Identify three special properties of water that make it unusual for a molecule of its.
Draw A Diagram Of Water Molecules Labeling The Hydrogen Bond And
The covalent bonds of water make it a polar molecule. Hydrogen bonds form between water molecules when the partially positive end of. Identify three special properties of water that make it unusual for a molecule of its.
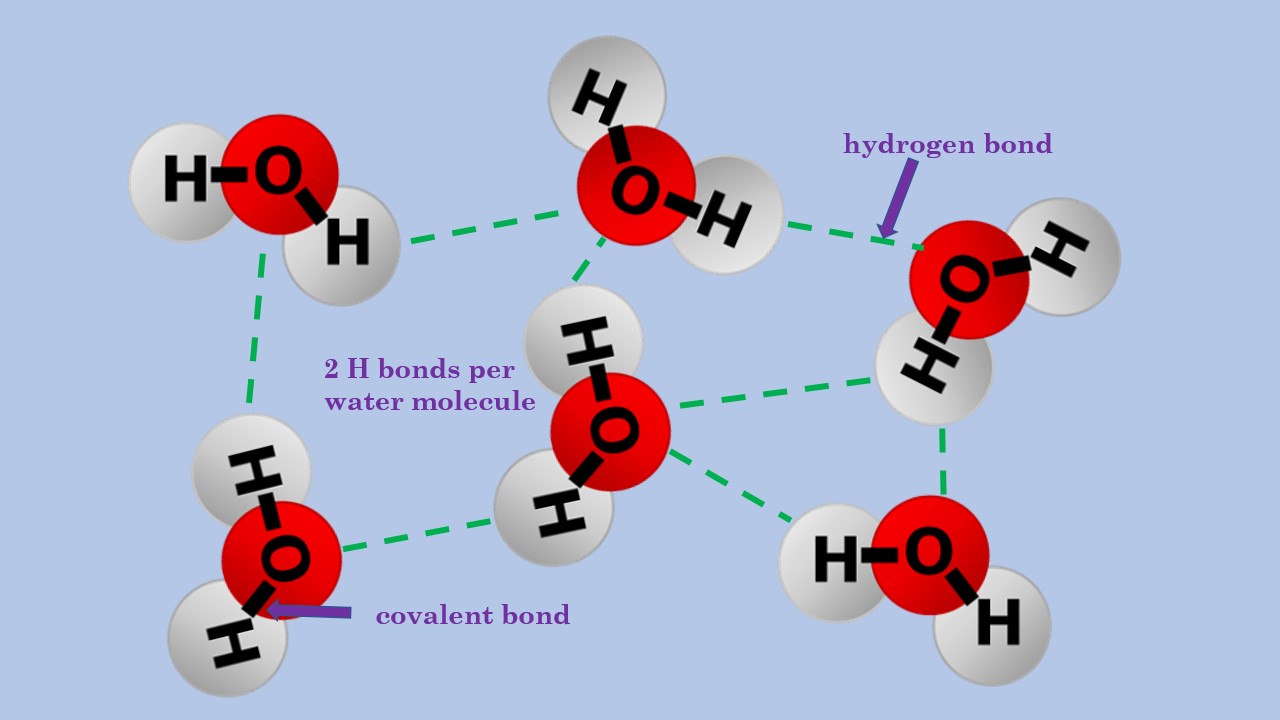
hydrogen bond between water molecules Diagram Quizlet
Hydrogen bonds form between water molecules when the partially positive end of. Identify three special properties of water that make it unusual for a molecule of its. The covalent bonds of water make it a polar molecule.
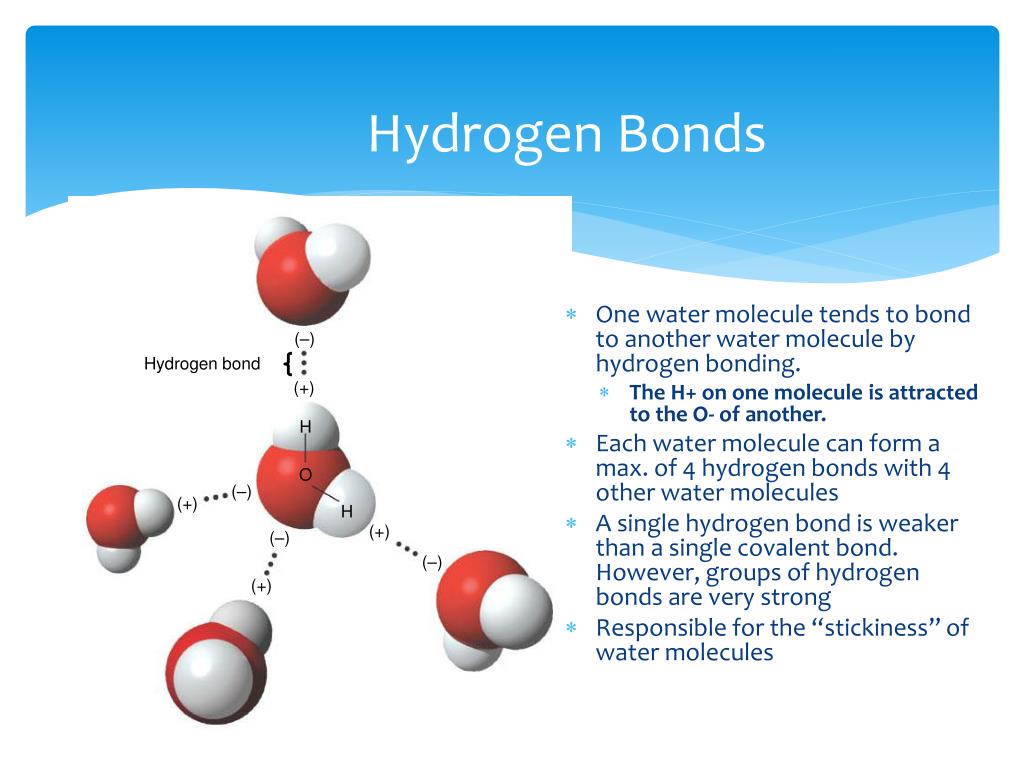
Are Hydrogen Bonds The Strongest Interactions Between Molecules?
Hydrogen bonds form between water molecules when the partially positive end of. The covalent bonds of water make it a polar molecule. Identify three special properties of water that make it unusual for a molecule of its.
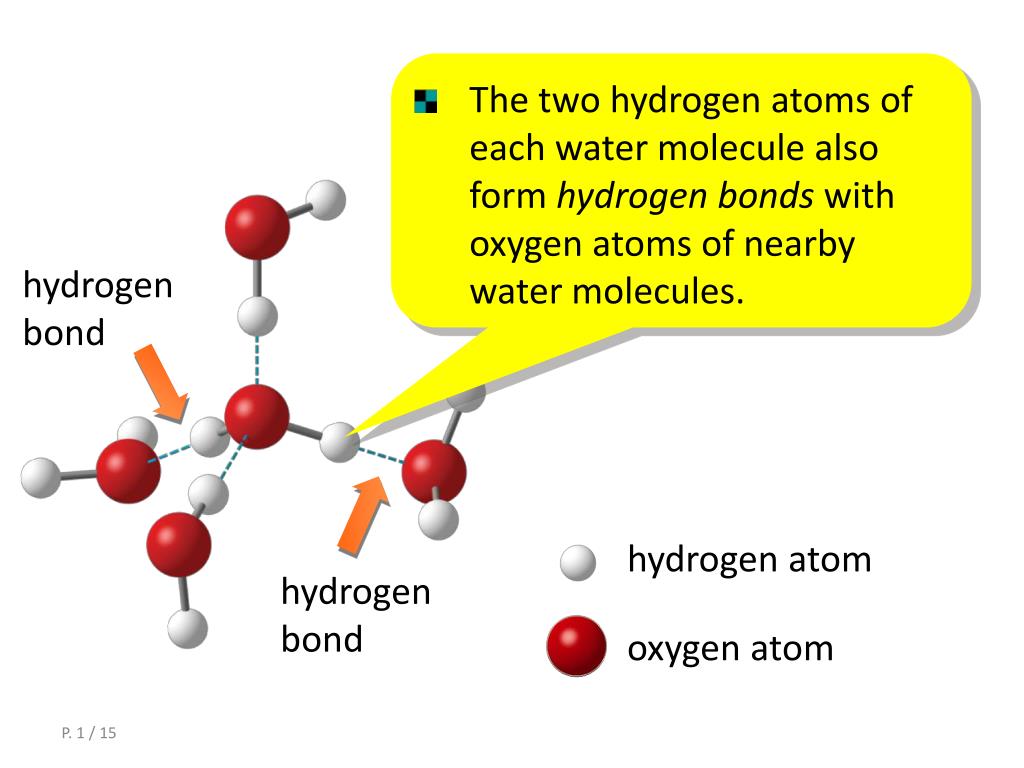
Diagram of hydrogen bonding between two water molecules Download
Hydrogen bonds form between water molecules when the partially positive end of. Identify three special properties of water that make it unusual for a molecule of its. The covalent bonds of water make it a polar molecule.
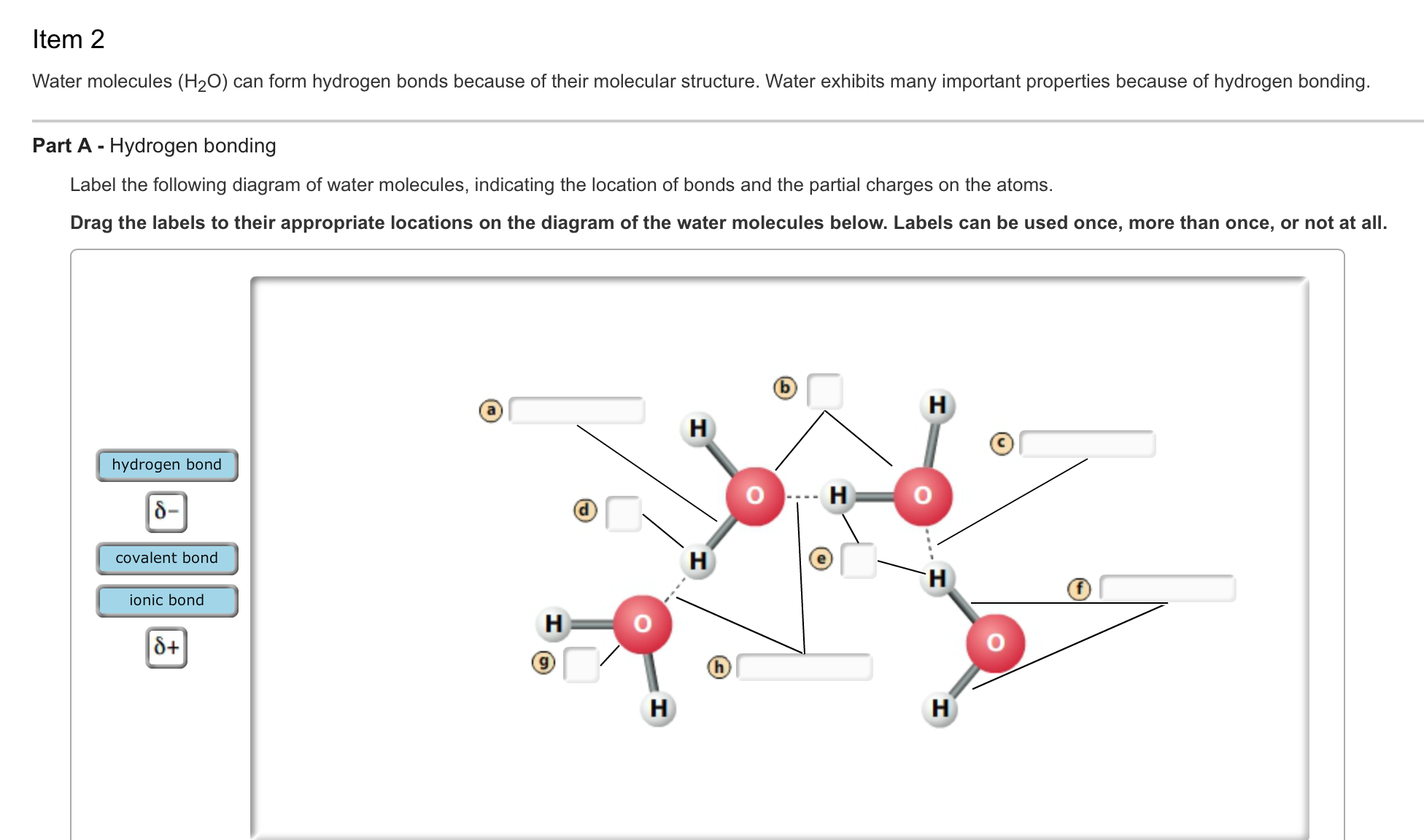
Solved Water molecules (H_2O) can form hydrogen bonds
The covalent bonds of water make it a polar molecule. Hydrogen bonds form between water molecules when the partially positive end of. Identify three special properties of water that make it unusual for a molecule of its.
Water Molecules Hydrogen Bonds
Identify three special properties of water that make it unusual for a molecule of its. Hydrogen bonds form between water molecules when the partially positive end of. The covalent bonds of water make it a polar molecule.
Draw A Hydrogen Bond Between Two Water Molecules
Identify three special properties of water that make it unusual for a molecule of its. Hydrogen bonds form between water molecules when the partially positive end of. The covalent bonds of water make it a polar molecule.
Water Molecules And Hydrogen Bonding Diagram
The covalent bonds of water make it a polar molecule. Identify three special properties of water that make it unusual for a molecule of its. Hydrogen bonds form between water molecules when the partially positive end of.
The Covalent Bonds Of Water Make It A Polar Molecule.
Hydrogen bonds form between water molecules when the partially positive end of. Identify three special properties of water that make it unusual for a molecule of its.